Quality Control and Media preparation laboratory
Quality Control and Media preparation laboratory (room no: 353, 361)
Quality assurance (QA) is an essential component of a clinical bacteriology laboratory. Laboratories practicing the principles of quality assurance generate reliable, relevant and cost-effective results, delivered timely and in the best interest of the patient.

National External Quality Assessment Scheme in Clinical Microbiology

1997-2022
Quality Control laboratory is the Proficiency Testing Provider (PTP) of the accredited ‘National External Quality Assessment Scheme in Microbiology’ conducted for the state and private sector microbiology laboratories in Sri Lanka. This program was granted accreditation to ISO 17043 standards as the PTP in 2021 by the Sri Lanka Accreditation Board (SLAB).
NEQAS in clinical microbiology was commenced in 1997 by Dr. R.S.B. Wickremasinghe and Dr. M. Atapattu, Consultant Bacteriologists worked in the Department of Bacteriology.
Objectives of the NEQAS program
- to improve the quality of medical laboratories in Sri Lanka conducting bacteriological investigations
- to standardize the performance of bacteriological techniques and antibiotic susceptibility testing
- to encourage use of standard reagents / methodology in bacterial identification and antimicrobial susceptibility testing (AST) to support quality control of in microbiology
- to monitor laboratory performance of clinical diagnostic microbiology
- to evaluate internal quality control measures
- to facilitate information exchange and continuing education among NEQAS laboratory Network
- stimulate performance improvement to ensure credibility of the laboratory
- to support the accreditation program of microbiology laboratories
Scope of Tests in clinical microbiology
- Gram stain interpretation
- Bacterial culture identification
- Antimicrobial Susceptibility Testing (AST) and result interpretation
Membership
Clinical microbiology laboratories in hospitals, universities or institutions in public or private sector, are eligible to request for enrollment in the program.
The participation is voluntary unless made mandatory by the Ministry of Health, Sri Lanka.
Laboratories are encouraged to enroll before the 30th of January each year. PT surveys for new member laboratories will be commenced only at the beginning of each year.
Enrollment is free for public sector hospitals and a nominal annual payment is charged from the private sector laboratories.
Contact details
Proficiency Testing Coordinator & Technical Advisor :
Dr. Lilani Karunanayake, Consultant Clinical Microbiologist
Technical Coordinator:
Mrs. A. Vayishnavi, senior Medical Laboratory Technologist
Postal Address:
Quality Control Laboratory, Department of Bacteriology
Medical Research Institute, Dr. Danister de Silva Mawatha,
Colombo 08, Sri Lanka
Telephone: 009411 269 3532 – 35 EXT: PTP laboratory – 353
Proficiency Testing Coordinator / Consultant – 332
Fax: 0094112691495 (Director office)
E mail: microneqas97.mri@gmail.com
Team members
Proficiency Testing Coordinator & Technical Advisor – Dr. Lilani Karunanayake, Consultant Clinical Microbiologist, Head, QC laboratory
Assistant Coordinator – Dr. P.H.P.D. Somaratne, Medical Officer
Technical Coordinator – Mrs. A. Vayishnavi, senior Medical Laboratory Technologist / Quality Control Laboratory
Medical Laboratory Technologist – Ms. K.D.D. Karavita
Health Laboratory Aide – Mr. D.N. Wijesinghe
Support staff – Mrs. G. Dilhara, Mrs. D. Ushetti
Quality Manager – Mrs. K.D.N. Gunawardena
Internal Auditor – Mrs. S. de Silva
Events
Workshop 2023 Theme: ‘Laboratory quality towards better patient care’
National Training Workshop on Clinical Microbiology for NEQAS participants – for Microbiologists and Medical Laboratory Technologists

Digital banner of the workshop
The 2-day program for 100 participants held on 16 & 17th February 2023 to mark the 25th Anniversary of the ‘National External Quality Assessment Scheme in (NEQAS) clinical microbiology’. The second edition of the ‘Handbook on National External Quality Assessment Scheme in Clinical Bacteriology’ was launched.
Further, Dr. Sudath Dharmaratne DDGLS/MOH awarded certificates to appreciate the laboratory staff for their dedication in achieving accreditation to the Clinical Bacteriology Laboratory and the NEQAs program. The event was sponsored by the WHO, Sri Lanka.










Award of certificates to the Clinical Bacteriology Laboratory staff for their dedication in achieving medical / clinical laboratory accreditation ISO 15189 and ISO 17043 as Proficiency Testing Provider. Certificates were presented by Dr. Sudath Dharmaratne, DDGLS, Ministry of Health, Sri Lanka.
CBL is the first and only government-owned microbiology laboratory to achieve accreditation status so far.


Laboratory visits 2016
Visited five microbiology laboratories in the Uva Province for on-site assessment

Laboratory visit to District General Hospital, Nuwara Eliya – with Dr. M. Abeywardene, Consultant Clinical Microbiologist and her team
Workshop 2018
One-Day National Training workshop for NEQAS laboratory network was held @ MRI auditorium with lectures and bench training.





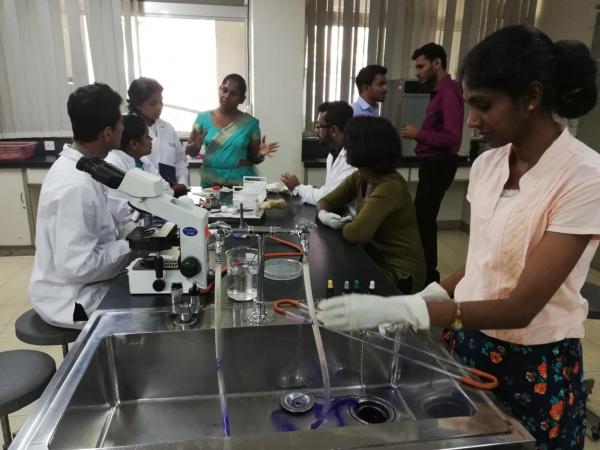





Workshop 2016
In November 2016, a one-day program was held on ‘Quality Assurance in Bacteriology for Medical Laboratory Technologists (MLT’s)’ of microbiology laboratories in both state- and private- sector institutions participating in NEQAS in Bacteriology, MRI. Workshop included lectures and demonstration of lab-based detection methods for antibiotic- resistant bacteria.

Participants in the one-day workshop at the Auditorium, MRI




